Abstract
Introduction
Since Core-binding factor acute myeloid leukemia (CBF-AML) is categorized as a favorable cytogenetic risk group, allogenic hematopoietic stem cell transplantation (HSCT) is generally not recommended during the first complete remission (CR1). However, approximately 30-50% of CBF-AML patients relapse, and about 50% of relapsed patients remain incurable. Therefore, in real-world clinical setting, the indication of HSCT even in CR1 is considered based on prognosis prediction and minimal residual disease (MRD) status. With respect to prognosis prediction by chromosomal abnormalities, the prognostic impact of loss of sex chromosome (LOS) in the patients with CBF-AML is still controversial. Previous studies suggest that the impact of LOS in predicting the prognosis of CBF-AML may depend on treatment strategies including HSCT. In addition, although CBF-AML develops LOS at a high rate of 20-40%, it is still unclear whether LOS is age-related or neoplastic.
Methods
Hokkaido Leukemia Net (HLN) is prospective cohort study collecting AML samples from hospitals of North Japan Hematology Study Group (NJHSG).Cases of CBF-AML that were registered in HLN from January 2010 to December 2019 were used for analysis in the present study. All the patients received either intensive or low-intensity chemotherapy. HSCT was performed at the discretion of the attending physicians even in CR1. Conventional cytogenetic analysis was performed at the time of diagnosis and after each treatment. FLT3-ITD and KIT exon17 mutations were analyzed using genome DNA template at the central laboratory. The results of genetic analysis were returned to physicians so that they could reflect the genetic result in their treatment choice. We evaluated the impacts of several factors including advanced age, poor response to chemotherapy, LOS, and KIT exon17 mutation on overall survival (OS) and relapse-free survival (RFS) in these patients. This study was conducted in compliance with ethical principles based on the Declaration of Helsinki and was approved by the institutional review board of Hokkaido University Hospital.
Results
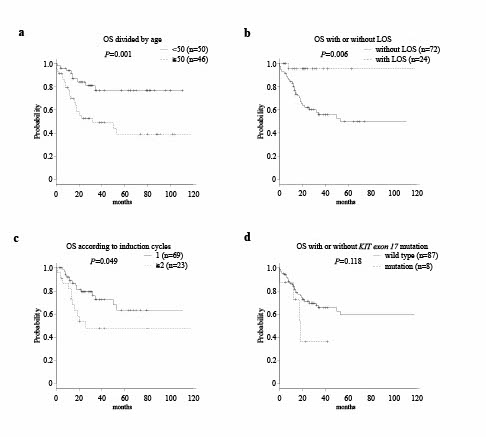
A total 96 CBF-AML patients including 62 patients with RUNX1/RUNX1T1 and 34 patients with CBFβ/MYH11 were enrolled in the present study. Intensive induction chemotherapy using '7+3' cytarabine plus anthracycline and low-intensity induction chemotherapy were performed in 90 patients and 6 patients, respectively. In 90 patients who underwent intensive induction chemotherapy, the CR rate after one cycle of induction therapy was 73.3%. Total CR rate as the best response was 91.7% for all CBF-AML patients. HSCT in CR1 was performed in 15 patients (15.6%) at the discretion of the attending physician based on genetic alterations, poor response to chemotherapy, and sustained detection of MRD after chemotherapy. There was no significant difference in 5-year OS (RUNX1/RUNX1T1 64.9% vs CBFβ/MYH11 52.9%; P=0.546) or 5-year RFS (63.6% vs 53.3%; P=0.364) between the CBF subtypes. LOS was significantly more frequent in patients with RUNX1/RUNX1T1 than in patients with CBFβ/MYH11 (35.5% vs 5.9%; P=0.001). Multivariate analyses for 5-year OS in CBF-AML patients revealed that age of 50 years or older (HR: 3.46, 95% CI: 1.47-8.11, P=0.004) and receiving 2 or more induction cycles (HR: 3.55, 95% CI: 1.57-8.05, P=0.002) were independently associated with worse OS and that LOS was independently associated with better OS (HR: 0.09, 95% CI: 0.01-0.71, P=0.022). We next sought to clarify whether LOS was age-related or neoplastic based on the transition of chromosome results. All the 24 patients with LOS achieved CR, and chromosome analysis at CR showed a normal karyotype in all of the 21 patients for whom chromosome analysis was performed. In the remaining 3 patients, only FISH analysis for RUNX1/RUNX1T1 or CBFβ/MYH11 was performed. Furthermore, chromosome analysis showed mosaics of metaphase cells with and without t(8;21) in 9 patients at diagnosis. In all of those patients, metaphase cells without t(8;21) showed a normal karyotype.
Conclusion
In our real-world clinical setting in which '7+3' induction was performed and the indication of HSCT was determined by prognostic factors and MRD status, LOS correlated to prolonged survival in CBF-AML patients. Furthermore, the transition of chromosome results revealed that LOS was not age-related physiological loss but part of neoplastic chromosomal abnormalities.
Teshima: CHUGAI PHARMACEUTICAL CO., LTD.: Research Funding; Janssen Pharmaceutical K.K.: Other; Novartis International AG: Membership on an entity's Board of Directors or advisory committees, Other, Research Funding; Gentium/Jazz Pharmaceuticals: Consultancy; Takeda Pharmaceutical Company: Honoraria, Membership on an entity's Board of Directors or advisory committees; Kyowa Kirin Co.,Ltd.: Honoraria, Research Funding; Astellas Pharma Inc.: Research Funding; Merck Sharp & Dohme: Membership on an entity's Board of Directors or advisory committees; TEIJIN PHARMA Limited: Research Funding; Nippon Shinyaku Co., Ltd.: Research Funding; Bristol Myers Squibb: Honoraria; Fuji pharma CO.,Ltd: Research Funding; Pfizer Inc.: Honoraria; Sanofi S.A.: Research Funding. Kondo: Astellas Pharma Inc.: Consultancy, Honoraria; Otsuka Pharmaceutical: Honoraria, Research Funding; Novartis Pharma KK: Honoraria; Bristol-Myers Squibb Company: Honoraria; Sumitomo Dainippon Pharma: Honoraria; Sanwa Kagaku Kenkyusho CO.,LTD: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal